 In the control arm, this applied to 32 % and 5 %, respectively. In the atezolizumab arm, 48 % of patients had any irAEs 11 % of which were grade 3-5. Each of these two groups was divided into patients with and without irAEs. Pooling of the three trials yielded a total of 2,503 patients who had been treated with either atezolizumab-containing regimens (n = 1,577) or control therapies (n = 926). IMpower130 and IMpower132 have assessed atezolizumab plus different chemotherapy regimens, while IMpower150 tested bevacizumab in addition to atezolizumab plus chemotherapy. 
evaluated the association between irAEs and OS in the IMpower130, IMpower132 and IMpower150 first-line trials. Based on this assumption, the post-hoc exploratory analysis presented by Socinski et al. Increasing evidence suggests that the occurrence of irAEs with PD-(L)1 inhibitor therapy might be predictive of improved outcomes. Immune-related adverse events (irAEs) have been reported in up to 80 % and 95 % of patients receiving checkpoint inhibitor monotherapy and combination therapy, respectively. In their summary, the authors noted that these updated results continue to support nivolumab/ipilimumab plus two cycles of chemotherapy as an efficacious first-line treatment option for patients with advanced NSCLC. After discontinuation, median duration of response was 14.5 months, and 56 % maintained their responses for ≥ 1 year. 
On the contrary, when compared indirectly to the total population randomized to combination treatment, these patients experienced improved survival with median OS of 27.5 months and a 24-month OS rate of 54 %. This showed that discontinuation did not have a negative impact on the long-term benefits. Most grade 3/4 treatment-related AEs (TRAEs) in the experimental arm emerged during the two chemotherapy cycles at the beginning of treatment.Ī post-hoc exploratory analysis assessed the outcomes of patients who had discontinued all components of nivolumab/ipilimumab plus chemotherapy due to TRAEs. No new safety signals were observed with longer follow-up. Similarly, patients treated in the experimental arm fared better with respect to OS in both non-squamous and squamous histology subgroups. The combination proved superior to chemotherapy in all PD-L1 expression categories (i.e., < 1 %, ≥ 1 %, ≥ 50 %) in terms of OS, PFS, and response ( Table 1). 5.6 months here, the 24-month rates amounted to 34 % vs. 8 % of patients in the experimental and control arms, respectively, were progression-free (HR, 0.67). Furthermore, PFS and response benefits were maintained with longer follow-up. Survival advantages occurred across all subgroups including patients with CNS metastases. įor overall survival, which constituted the primary endpoint, the analysis showed durable efficacy of the combination. 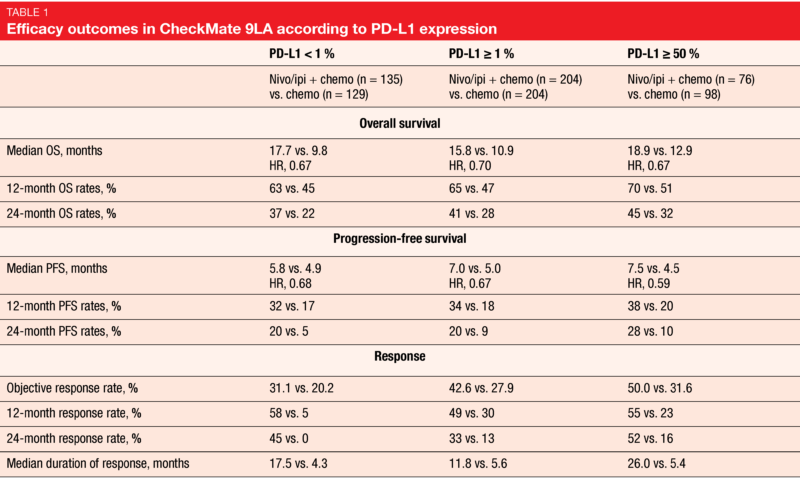
reported updated efficacy and safety findings after a minimum follow-up of 2 years, as well as outcomes in patients who discontinued treatment due to adverse events. CheckMate 9LA included approximately 360 patients with stage IV or recurrent disease in each arm and demonstrated significant OS, PFS, and ORR improvements with the immunotherapy-based regimen compared to four cycles of standard chemotherapy. Based on the randomized, phase III CheckMate 9LA study, the first-line regimen of nivolumab plus ipilimumab and two cycles of chemotherapy has been approved in the indication of metastatic NSCLC without EGFR or ALK aberrations in many countries. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
